L. Sobczyk, D. Chudoba, P.M. Tolstoy, A. Filarowski
“Some Brief Notes on Theoretical and Experimental Investigations of Intramolecular Hydrogen Bonding”
Molecules, 2016, 21, 1657-1/19
DOI:10.3390/molecules21121657
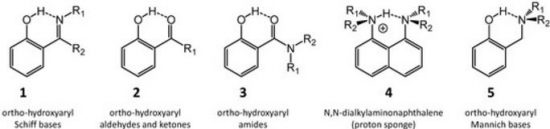
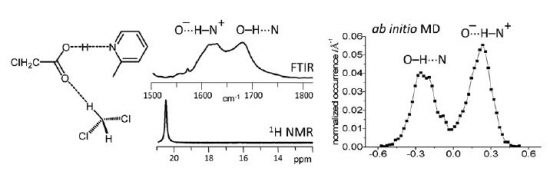
A review of selected literature data related to intramolecular hydrogen bonding in ortho-hydroxyaryl Schiff bases, ortho-hydroxyaryl ketones, ortho-hydroxyaryl amides, proton sponges and ortho-hydroxyaryl Mannich bases is presented. The paper reports on the application of experimental spectroscopic measurements (IR and NMR) and quantum-mechanical calculations for investigations of the proton transfer processes, the potential energy curves, tautomeric equilibrium, aromaticity etc. Finally, the equilibrium between the intra- and inter-molecular hydrogen bonds in amides is discussed.