A. Lupachev, E. Abakumov, S. Gubin
“The Influence of Cryogenic Mass Exchange on the Composition and Stabilization Rate of Soil Organic Matter in Cryosols of the Kolyma Lowland (North Yakutia, Russia)”
Geosciences, 2017, 7, 24
DOI:10.3390/geosciences7020024
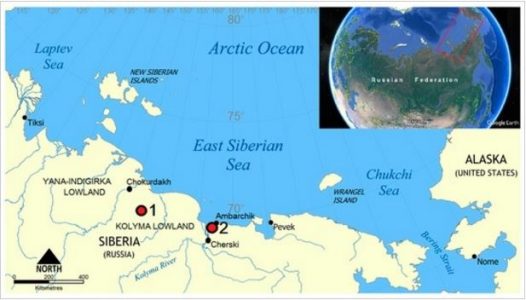
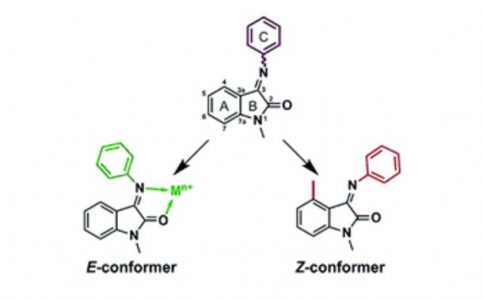
Soil organic matter (SOM) was studied in different types of organo-mineral material (from surface horizons and partially isolated materials—cryoturbated or buried horizons) sampled from the surface horizons, the central parts of the Cryosol profiles, and the lower active layer. We found that the humic acids (HAs) of the cryoturbated and buried horizons showed an increased degree of oxidation and an increment of alkylaromatic and protonized aromatic fraction content. In contrast, the HAs of the surface horizons showed increased values of alkylic carbon components. The content of free radicals was essentially higher in the surface layers than in the cryoturbated and buried layers. While the bulk soil organic matter composition (total organic carbon, total nitrogen, and aromatic/aliphatic values) was not essentially different between surface, cryoturbated, and buried horizons, there were essential differences in elemental composition, carbon species, and free radical content. This indicates that the degree of humification in cryoturbated and buried organo-mineral material is higher than in surface horizons and that partial isolation results in relative stabilization of such material in soil profiles.