 Sergey Smirnov and Alexander Ivanov have completed the training course “High Resolution NMR Spectrometer Service and Maintenance” in Bruker, Switzerland, and got the certificates.
Sergey Smirnov and Alexander Ivanov have completed the training course “High Resolution NMR Spectrometer Service and Maintenance” in Bruker, Switzerland, and got the certificates.
 Sergey Smirnov and Alexander Ivanov have completed the training course “High Resolution NMR Spectrometer Service and Maintenance” in Bruker, Switzerland, and got the certificates.
Sergey Smirnov and Alexander Ivanov have completed the training course “High Resolution NMR Spectrometer Service and Maintenance” in Bruker, Switzerland, and got the certificates.
In the VII Symposium «Nuclear Magnetic Resonance in Chemistry, Physics and Biological Sciences» that took place on 25-28 of September 2012 in Warsaw, Poland, we presented a talk:
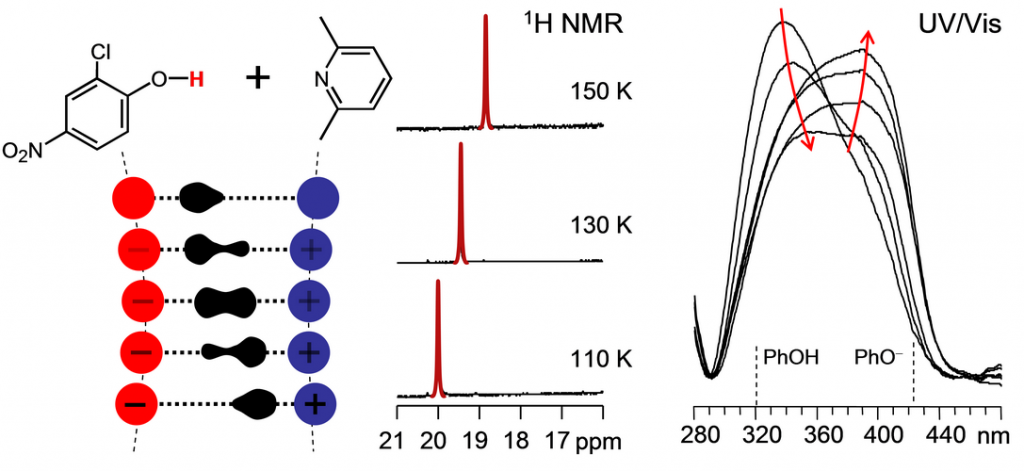
Peter Tolstoy, Benjamin Koeppe, Jing Guo, Svetlana Pylaeva, Elena Tupikina, Gleb Denisov, Hans-Heinrich Limbach
«Effects of Solvation Shell Structure and Dynamics on Geometry and NMR/UV-vis Spectra of H-bonded Complexes»
and two posters:
1) Elena Tupikina, Peter Tolstoy, Sergey Smirnov, Nikolai Golubev, Gleb Denisov «Hydroxyphenyl Benzimidazole as Catalyst for Hydrolysis Reaction: Serine Protease Active Site Model»
2) Svetlana Pylaeva, Peter Tolstoy, Benjamin Koeppe, Christoph Allolio, Daniel Sebastiani, Gleb Denisov «Distribution of Hydrogen Bond Geometries and Spectroscopic Parameters Due to the Solvent-Solute Interactions Studied by Means of Computer Simulations».
B. C. K. Ip, I. G. Shenderovich, P. M. Tolstoy, J. Frydel, G. S. Denisov, G. Buntkowsky and H.-H. Limbach,
“NMR Studies of Solid Pentachlorophenol-4-Methylpyridine Complexes Exhibiting Strong OHN Hydrogen Bonds: Geometric H/D Isotope Effects and Hydrogen Bond Coupling Cause Isotopic Polymorphism”
J. Phys. Chem. A 2012, ASAP.
DOI: 10.1021/jp305863n.
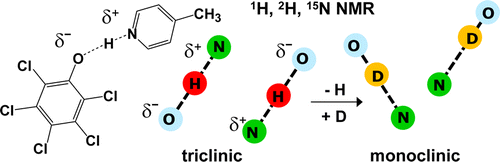
We have studied the hydrogen bond interactions of 15N labeled 4-methylpyridine (4-MP) with pentachlorophenol (PCP) in the solid state and in polar solution using various NMR techniques. Read more
On the VVV-2012 conference that took place in Novosibirsk on the 15-20 of July 2012 was presented a talk
P. M. Tolstoy, B. Koeppe, J. Guo, S. A. Pylaeva, G. S. Denisov, H.-H. Limbach, “Reaction Pathways of Proton Transfer in Anionic OHO Hydrogen Bonded Complexes”.
On the NMRCM-2012 conference that took place in St. Petersburg on the 9-14 of July 2012 were presented two talks
P. Tolstoy, B. Koeppe, S. Pylaeva, E. T. J. Nibbering, D. Sebastiani, G. Denisov, H.-H. Limbach, “Solvation of H-Bonded Complexes by Polar Aprotic Solvents: Coupling of the Proton Position to the Solvent Configuration”.
and
S. Pylaeva, P. Tolstoy, B. Koeppe, C. Allolio, D. Sebastiani, G. Denisov, “Fluctuations of H-bond geometry due to the solvent-solute interactions studied by means of computer simulations”.
The latter was awarded for the best poster.
J. Guo, P. M. Tolstoy, B. Koeppe, N. S. Golubev, G. S. Denisov, S. N. Smirnov, H.-H. Limbach
“Hydrogen Bond Geometries and Proton Tautomerism of Homo-Conjugated Anions of Carboxylic Acids Studied via H/D Isotope Effects on 13C NMR Chemical Shifts”
J. Phys. Chem. A 2012, ASAP.
DOI: 10.1021/jp304943h.
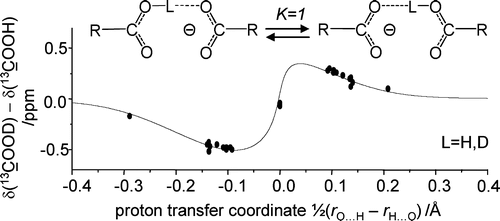
Ten formally symmetric anionic OHO hydrogen bonded complexes, modeling Asp/Glu amino acid side chain interactions in nonaqueous environment (CDF3/CDF2Cl solution, 200–110 K) have been studied by 1H, 2H, and 13C NMR spectroscopy Read more
Gurinov, A. A.; Mauder, D.; Akcakayiran, D.; Findenegg, G. H.; Shenderovich, I. G.
“Does Water Affect the Acidity of Surfaces? The Proton-Donating Ability of Silanol and Carboxylic Acid Groups at Mesoporous Silica.”
ChemPhysChem 2012, 13 (9), 2282-2285.
DOI: 10.1002/cphc.201200204
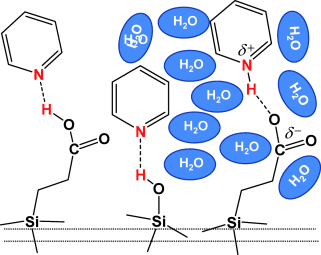
A study of the influence of water on the effective acidity of silanol and carboxylic acid groups of propionic acid functionalized SBA-15 reveals that to affect the proton-donating ability of an acidic group at the surface, water should be able to form a solvation shell around that group. As a result, water does not affect the acidity of native SBA-15 but dramatically enhances that of SBA-15 functionalized with propionic acid moieties.
 Russian-Finnish Conference on Technology Transfer, Entrepreneurship and Research Infrastructure Management took place in Aalto university, Finland.
Russian-Finnish Conference on Technology Transfer, Entrepreneurship and Research Infrastructure Management took place in Aalto university, Finland.
Key themes at Conference:
 Gorobets, N. Y.; Yermolayev, S. A.; Gurley, T.; Gurinov, A. A.; Tolstoy, P. M.; Shenderovich, I. G.; Leadbeater, N. E.
Gorobets, N. Y.; Yermolayev, S. A.; Gurley, T.; Gurinov, A. A.; Tolstoy, P. M.; Shenderovich, I. G.; Leadbeater, N. E.
"Difference between 1H NMR signals of primary amide protons as a simple spectral index of the amide intramolecular hydrogen bond strength."
J. Phys. Org. Chem. 2011, 25 (4), 287-295.
DOI: 10.1002/poc.1910
The effect of the intramolecular H-bonding of the primary amide group on the spectral properties and reactivity of this group towards electrophiles has been studied in systematic rows of 1,2,5,6,7,8-hexahydro-7,7-dimethyl-2,5-dioxo-1-R-quinoline-3-carboxamides and 2-aryliminocoumarin-3-carboxamides using 1H and 15N NMR spectroscopy and the kinetics of model reactions. Read more