Archive for A. Grevtsev
J Org. Chem. 2015, 80, 18-29
M.S. Novikov, A.F. Khlebnikov, N.V. Rostovskii, S.Tcyrulnikov, A.A. Suhanova, K.V. Zavyalov, D.S. Yufit
“Pseudopericyclic 1,5- versus Pericyclic 1,4- and 1,6-Electrocyclization in Electron-Poor 4‑Aryl-2-azabuta-1,3-dienes: Indole Synthesis from 2H‑Azirines and Diazo Compounds”
J Org. Chem., 2015, 80, 18-29
DOI: 10.1021/jo501051n
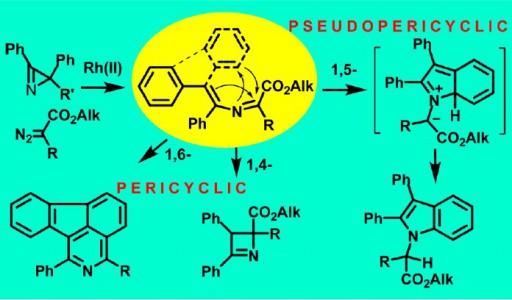
Transformations of 2-azabuta-1,3-dienes, formed in Rh2(OAc)4-catalyzed reactions of diazo carbonyl compounds with 2H-azirines, dramatically depend on the nature of substituents. 4,4-Diphenyl-2-azabuta-1,3-dienes with two electron- acceptor substituents at C1 undergo thermal 1,5-cyclization to give indoles in good yields.
The increase in electronwithdrawing ability of C1-substituents facilitates the reaction that proceeds via pseudopericyclic 1,5-electrocyclization of 2- azabutadiene into 7aH-indolium ylide followed by prototropic shift. 3,4-Diphenyl-2-azabuta-1,3-dienes, resulting from reaction of 2,3-diphenyl-2H-azirine and diazo compounds, do not produce indoles via 1,5-cyclization due to the trans-configuration of the 4-Ph-group and the nitrogen, but undergo 1,4-cyclization to 2,3- dihydroazetes. 1,6-Cyclization into 2H-1,4-oxazines with participation of the oxygen of ester or amide group at C1 of corresponding 2-azabuta-1,3-dienes does not take place due to kinetic and thermodynamic reasons. Instead of this, 1,6-electrocyclization with participation of phenyl substituent at C4 of the 2-azabuta-1,3- dienes, providing isoquinoline derivatives, can occur at elevated temperatures. The DFT-calculations (mPWB1K/6-31+G(d,p)) confirm the dependence of 2-azabuta-1,3-diene transformation type on the nature of substituents.
December
Total in october 1504 service applications were carried out.
All together measured:
- 1420 1H spectra
- 407 13C spectra
- 238 DEPT spectra
- 23 COSY spectra
- 11 NOESY spectra
- 38 31P spectra
- 75 19F spectra
153 applications were carried out which jointly took 1544 hours of measurements.
PCCP 2015
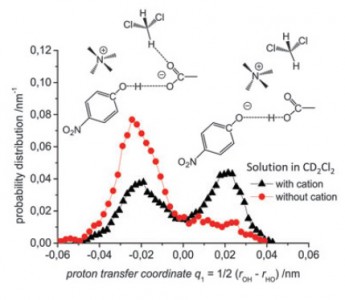
S. Pylaeva, C. Allolio, B. Koeppe, G.S. Denisov, H.-H. Limbach, D. Sebastiani, P.M. Tolstoy
“Proton transfer in hydrogen bonded complex caused by solvation shells fluctuations: ab initio MD study of anionic phenolate-carboxylic acid and neutral pyridine-carboxylic acid systems”
PCCP, 2015, accepted
DOI: 10.1039/C4CP04727C
Chem. Heterocycl. Compd., 2015, 50(11)
A. Ya. Bespalov, T. L. Gorchakova, A. Yu. Ivanov, M. A. Kuznetsov, L. M. Kuznetsova, A. S. Pankova, L. I. Prokopenko, M. S. Avdontceva
«Alkylation and Aminomethylation of 1,3-Dihydro-2Н-Benzimidazole-2-Thione»
Chem. Heterocycl. Compd, 2015, 50(11), ASAP
DOI: 10.1039/c4ra07840c
Alkylation of 1,3-dihydro-2Н-benzimidazole-2-thione (2-mercapto benzimidazole) with bromoethane and chloroacetic acid derivatives occurrs at the sulfur atom, leading to the corresponding 2-sulfanylbenz-imidazole derivatives. Aminomethylation of 1,3-dihydro-2Н-benzimidazole-2-thione with piperidine and 4-methylpiperidine gives reaction products at both nitrogen atoms, while reaction with morpholine gives derivative at only one nitrogen atom, which is in an equilibrium with the starting compound and bis-adduct in DMSO solution
RSC Advances, 2014
A.S. Pankova, M.A. Samartsev, I.A. Shulgin, P.R. Golubev, М.А. Кuznetsov
“Synthesis of thiazolidines via regioselective addition of unsymmetric thioureas to maleic acid derivatives”
RSC Advances, 2014, 4, 51780-51786
DOI: 10.1039/c4ra07840c
Wide range of unsymmetric thioureas has been studied in reaction with N-arylmaleimides and maleic anhydride. The regioselectivity of the addition depends not only on steric factors but on both solvent polarity and type of maleic acid derivative (imide or anhydride). The general regularities have been established providing practical guidelines to control the reaction result. The unequivocal structural assignment of all products has been done using NMR spectroscopy including 15N–1H HMBC experiments.
Visitors from the University of Tokyo
Today prof. Kaoru Yamanouchi and prof. Kiichiro Hotayama from the University of Tokyo have visited the Center.
J. Phys. Chem. A, 2014
E.Yu. Tupikina, G.S. Denisov, P.M. Tolstoy
“NMR Study of CHN Hydrogen Bond and Proton Transfer in 1,1-dinitroethane Complex with 2,4,6-trimethylpyridine”
J. Phys. Chem. A., 2014, accepted
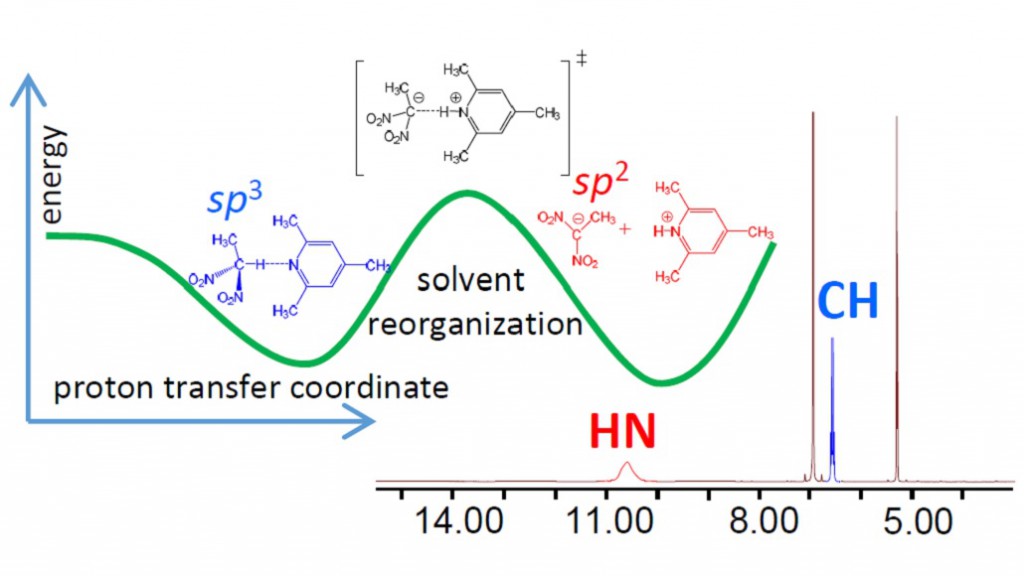
Intermolecular complex with CHN hydrogen bond formed by 1,1-dinitroethane (DNE) and 2,4,6-trimethylpyridine (collidine) dissolved in CD2Cl2 was studied experimentally by 1H NMR spectroscopy at 180-300 K. Equilibrium between molecular CH⋅⋅⋅N form and zwitterionic C−/HN+ form was detected in slow exchange regime in NMR time scale. No sign of direct C−⋅⋅⋅HN+ bond was observed; the ion pair is likely to be held by Coulomb interactions. Moreover, there are indications that the protonated base is involved in formation of homo-conjugated (NHN)+ collidine-collidinium hydrogen bonded complexes.
The reaction pathway of proton transfer in DNE-pyiridine complex in vacuum was studied computationally at B3LYP/6-31++G(d,p) level of theory. NMR chemical shifts and coupling constants were calculated for a series of snapshots along the proton transfer coordinate. While central carbon atom has pyramidal (sp3) configuration in DNE, it is flat (sp2) in DNE carbanion. As a result, the most indicative computed NMR parameter reflecting hybridization of carbon atom appeared to be 1JCC, which starts to change rapidly as soon as structure with quasi-symmetric C⋅⋅H⋅⋅N bond is reached. Couplings within the hydrogen bridge, 1JCH, 1hJHN and 2JCN, can serve as good indicators of the degree of proton transfer.
Chem. Eur. J., 2014
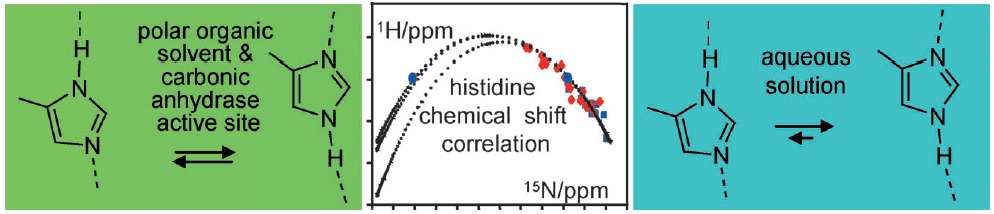
I.G. Shenderovich, S.B. Lesnichin, C. Tu, D.N. Silverman, P.M. Tolstoy, G.S. Denisov, H.-H. Limbach
“NMR Studies of Active Site Properties of Human Carbonic Anhydrase II using 15N labeled 4-Methylimidazole as a Local Probe and Histidine Hydrogen Bond Correlations”
Chem. Eur. J., 2014, accepted
DOI: 10.1002/chem.201404083
By using a combination of liquid and solid-state NMR spectroscopy, 15N-labeled 4-methylimidazole (4-MI) as a local probe of the environment has been studied: 1) in the polar, wet Freon CDF3/CDF2Cl down to 130 K, 2) in water at pH 12, and 3) in solid samples of the mutant H64A of human carbonic anhydrase II (HCA II). In the latter, the active-site His64 residue is replaced by alanine; the catalytic activity is, however, rescued by the presence of 4-MI. For the Freon solution, it is demonstrated that addition of water molecules not only catalyzes proton tautomerism but also lifts its quasidegeneracy. The possible hydrogen-bond clusters formed and the mechanism of the automerism are discussed. Information about the imidazole hydrogen-bond geometries is obtained by establishing a correlation between published 1H and 15N chemical shifts of the imidazole rings of histidines in proteins. This correlation is useful to distinguish histidines embedded in the interior of proteins and those at the surface, embedded in water. Moreover, evidence is obtained that the hydrogen-bond geometries of His64 in the active site of HCA II and of 4-MI in H64A HCA II are similar. Finally, the degeneracy of the rapid tautomerism of the neutral imidazole ring His64 reported by Shimahara et al. (J. Biol. Chem. 2007, 282, 9646) can be explained with a wet, polar, nonaqueous active-site conformation in the inward conformation, similar to the properties of 4-MI in the Freon solution. The biological implications for the enzyme mechanism are discussed.
Spinus-2014
Our contribution to the 11th Winter School conference “Magnetic Resonance and its applications”, Spinus-2014 (title of report in Russian):
1. Толстой П. М., Чан-Хут М., Шазад Ш., Тони М., Шендерович И. Г., Денисов Г. С., Лимбах Х. Х., «Исследование кооперативности водородных связей в витамин B6-зависимых ферментах и модельных системах методом ЯМР»
2. Кукушкина Е.В., Шеляпина М.Г., «ЯМР 51V в неупорядоченных сплавах Fe(1-x)V(x) и YFe(12-x)V(x)»