As part of the course “Experimental methods in NMR” students of Physics Department have reviewed a modern scientific equipment in our center.
Archive for March 30, 2015
Students of Physics Department
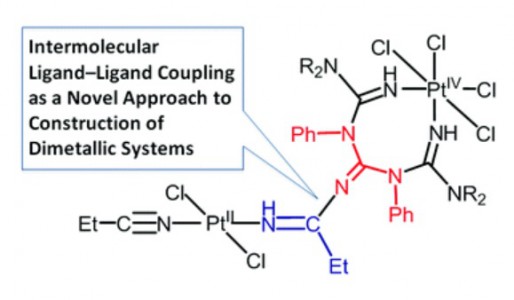
Dalton Trans. 2015, 44, 6003-6011
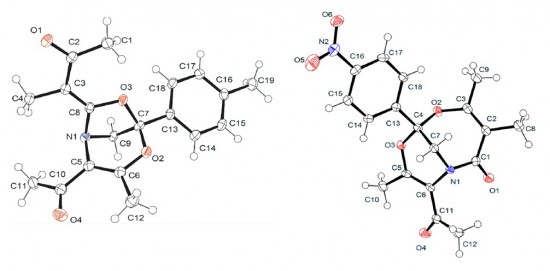
T.V. Serebryanskaya, A.S. Novikov, P.V. Gushchin, A.A. Zolotarev, V.V. Gurzhiy, V.Yu. Kukushkin
“Coupling of platinated triguanides with platinumactivated nitriles as a novel strategy for generation of dimetallic systems”
Dalton Trans., 2015, 44, 6003-6011 DOI:10.1039/c4dt03870c
One of two PtIV-activated propanenitriles in trans-[PtCl4(EtCN)2] is involved in platinum(IV)-mediated nitrile–imine coupling with the platinum(II)-based metallacycles [PtCl2{NH[double bond, length as m-dash]C(NR2)N(Ph)C([double bond, length as m-dash]NH)N(Ph)C(NR2)[double bond, length as m-dash]NH}] [R2 = Me2 (1a), C5H10 (1b)] yielding diplatinum products, whose structures depend on molar ratios between the reactants. At a 1:1 ratio, the mixed-valence platinum(II)/platinum(IV) species [PtCl4{NH[double bond, length as m-dash]C(NR2)N(Ph)C{[double bond, length as m-dash][(N(Et)C[double bond, length as m-dash]NH)PtCl2(EtCN)]}N(Ph)C(NR2)[double bond, length as m-dash]NH}] [R2 = Me2 (2a), (CH2)5 (2b)] were generated, whereas at a 1:2 ratio the dinuclear platinum(II)/platinum(II) complexes [PtCl2{NH[double bond, length as m-dash]C(NR2)N(Ph)C{[double bond, length as m-dash][(N(Et)C[double bond, length as m-dash]NH)PtCl2(EtCN)]}N(Ph)C(NR2)[double bond, length as m-dash]NH}] [R2 = Me2 (3a), (CH2)5 (3b)] were obtained. In contrast to the nitrile–imine coupling observed for the platinum(IV) dinitrile complex, the reaction between the platinum(II) congener trans-[PtCl2(EtCN)2] and any one of 1a,b gives exclusively the substituted dimetallic platinum(II)/platinum(II) products [PtCl2{NH[double bond, length as m-dash]C(NR2)N(Ph)C{[double bond, length as m-dash][(NH)PtCl2(EtCN)]}N(Ph)C(NR2)[double bond, length as m-dash]NH}] [R2 = Me2 (6a), (CH2)5 (6b)] featuring platinum-containing guanidine 1 as one of the ligands. Complexes 2a,b, 3a,b, and 6a,b were characterized by elemental analyses (C, H, N), HRESI-MS, IR, 1H NMR spectroscopy, and DTA/TG. The molecular and crystal structure of 2a·2CDCl3 was additionally studied by single-crystal X-ray diffraction. Complexes 2a,b undergo further redox transformation in solutions, and single crystals of [PtCl2{NH[double bond, length as m-dash]C(NMe2)N(Ph)C{[double bond, length as m-dash][(N(Et)C[double bond, length as m-dash]NH)PtCl2(MeCN)]}N(Ph)C(NMe2)[double bond, length as m-dash]NH}]·2CH2Cl2 (3′a·2CH2Cl2) were obtained from 2a in a CH2Cl2–MeCN–C2H4Cl2 mixture and studied by X-ray crystallography. The driving forces for the generation of diplatinum products 2 and 3 were elucidated based on a quantum-chemical study.
British J. App. Sci. Technol. 2015
A. Stepakov, S. Galkina, D. Bogomaz, E. Gaginskaya, A. Saifitdinova
“Modified Synthesis of 6-carboxyfluorescein (6-FAM): Application to Probe Labeling for Conventional Cytogenetics”
British J. App. Sci. Technol., 2015, 7, 423-428
DOI:10.9734/BJAST/2015/15991
Aims: Fluorescent in situ hybridization (FISH), the routine technique of molecular cytogenetics, is widely used to detect and localize the presence of specific nucleic acids sequences in chromosomes, cell nucleus space, cells and tissue samples through the use of highly complementary probes to targets sequence. Expansion of FISH method application for research purposes and medical diagnostics requires efficient and low-cost production of labeled nucleic acid probes.
Methodology and Results: We developed an effective method of fluorescein hydroxyalkyl carboxamides synthesis. This modification of the basic protocol of 6-carboxyfluorescein (6-FAM) synthesis enabled the production of highly reactive conjugate perfectly suitable for terminal labeling of newly generated oligonucleotides. Efficiency of 6-FAM labeled oligonucleotides obtained by the use of modified protocol has been proved for conventional cytogenetics.
Conclusion: The suggested procedure of 6-FAM labeled oligonucleotides synthesis allows obtaining the high yield of directly labeled FISH probes. The introduction of this method into practice of cytogenetic studies will improve their efficiency and reduce the cost of an examination.
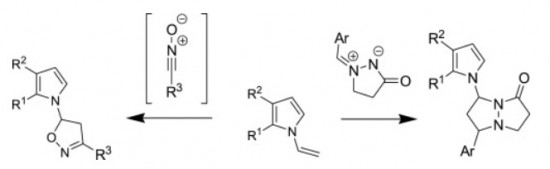
Tetrahedron 2015 (71) 2071-2078
M.M. Efremova, A.P. Molchanov, A.V. Stepakov, R.R. Kostikov, V.S. Shcherbakova, A.V. Ivanov
“A highly efficient [3+2] cycloaddition of nitrile oxides and azomethine imines to N-vinylpyrroles”
Tetrahedron, 2015, 71, 2071-2078
DOI:10.1016/j.tet.2015.02.058
1,3-Dipolar cycloaddition of various nitrile oxides to substituted N-vinylpyrroles proceed with high efficiency and selectivity with the formation of single isomer of 5-pyrrolyl-substituted isoxazoline. The reaction of N-vinylpyrroles with cyclic azomethine imines occurs regioselectively affording 7-(pyrrol-1-yl)- substituted pyrazolo[1,2-a]pyrazolones as a mixture of two diastereomers.
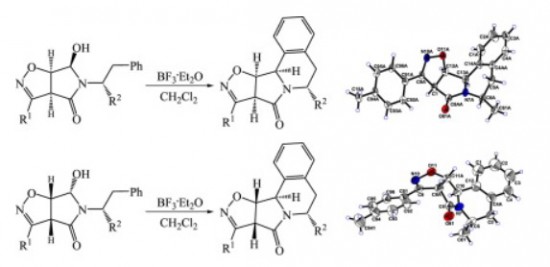
Tetrahedron 2015 (71) 1952-1958
M.S. Ledovskaya, A.P. Molchanov, V.M. Boitsov, R.R. Kostikov, A.V. Stepakov
“An efficient synthesis of substituted isoxazolopyrroloisoquinolines via diastereoselective N-acyliminium ion cyclization”
Tetrahedron, 2015, 71, 1952-1958
DOI:10.1016/j.tet.2015.02.031
A simple and efficient strategy was developed for the synthesis of fused pyrrolo[2,1-a]isoquinoline ring systems. The 5- and 6-substituted isoxazolopyrroloisoquinolines were readily prepared via diastereoselective N-acyliminium ion cyclization of 5-(1-R(or 2-R)-substituted-2-phenylethyl)-6-hydroxytetrahydro-4H-pyrrolo[3,4-d]isoxazol-4-ones derived from the corresponding bicyclic dihydroisoxazoles.
European Journal of Inorganic Chemistry
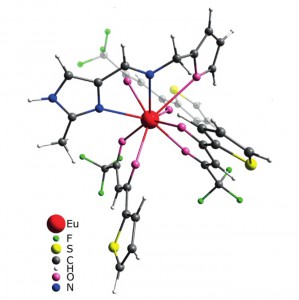
K.S. Kisel, G. Linti, G.L. Starova, V.V. Sizov, A.S. Melnikov, A.P. Pushkarev, M.N. Bochkarev, E.V. Grachova, S.P. Tunik
“Syntheses, Structures, and Photophysical Properties of Eu and Lu Diketonates with a Neutral Polydentate Imidazolylmethanamine Ligand”
Eur. J. Inorg. Chem., 2015
DOI: 10.1002/ejic.201403186
The Schiff base NNO ligand 1-(furan-2-yl)-N-[(2-methyl-1H-imidazol-4-yl)methylene]methanamine was synthesized and structurally characterized by XRD crystallography, mass spectrometry, and NMR spectroscopy. Quantum-chemical calculations revealed conformational flexibility of the ligand backbone to give two different conformations with nearly equal ground-state energies. The orientation of two nitrogen atoms and the oxygen atom in one conformation is a good fit for the NNO tridentate coordination mode, whereas the other would allow the NN coordination mode only. Two lanthanide complexes [Ln(tta)3(NNO)] (Ln = EuIII and LuIII; tta = thenoyltrifluoroacetone) were prepared and studied spectroscopically. The structures of the complexes were optimized by the DFT approach. The NNO ligand in the Eu complex displays tridentate NNO coordination, whereas the ligand is only NN-coordinated in the Lu complex. The Eu complex shows bright red metal-centered phosphorescence under excitation into the ligand (π–π*) absorption bands with a quantum yield of ca. 80 % and a lifetime of 580 μs. A mechanism for energy transfer between the ligands and metal centers was suggested and confirmed by DFT and time-dependent DFT (TDDFT) studies. An organic light-emitting diode (OLED) device based on the Eu complex incorporated into a poly(9-vinylcarbazole) (PVK) matrix was prepared. A study of the characteristics of the device revealed electrochromism of the system owing to variations in the efficacy of metal-centered and matrix emission at different strengths of applied electric field.
Beilstein J. Org. Chem. 2015
N.V. Rostovskii, M.S. Novikov, A.F. Khlebnikov, G.L. Starova, M.S. Avdontseva
“Azirinium ylides from α-diazoketones and 2H-azirines on the route to 2H-1,4-oxazines: three-membered ring opening vs 1,5-cyclization”
Beilstein J. Org. Chem., 2015, 11, 302-312
DOI: 10.3762/bjoc.11.35
Strained azirinium ylides derived from 2H-azirines and α-diazoketones under Rh(II)-catalysis can undergo either irreversible ring opening across the N–C2 bond to 2-azabuta-1,3-dienes that further cyclize to 2H-1,4-oxazines or reversibly undergo a 1,5-cyclization to dihydroazireno[2,1-b]oxazoles. Dihydroazireno[2,1-b]oxazoles derived from 3-aryl-2H-azirines and 3-diazoacetylacetone or ethyl diazoacetoacetate are able to cycloadd to acetyl(methyl)ketene generated from 3-diazoacetylacetone under Rh(II) catalysis to give 4,6-dioxa-1-azabicyclo[3.2.1]oct-2-ene and/or 5,7-dioxa-1-azabicyclo[4.3.1]deca-3,8-diene-2-one derivatives. According to DFT calculations (B3LYP/6-31+G(d,p)), the cycloaddition can involve two modes of nucleophilic attack of the dihydroazireno[2,1-b]oxazole intermediate on acetyl(methyl)ketene followed by aziridine ring opening into atropoisomeric oxazolium betaines and cyclization. Azirinium ylides generated from 2,3-di- and 2,2,3-triaryl-substituted azirines give rise to only 2-azabuta-1,3-dienes and/or 2H-1,4-oxazines.
Bowling Green State University Visitors
February
Total in February 1331 service applications were carried out.
All together measured:
- 1268 1H spectra
- 265 13C spectra
- 137 DEPT spectra
- 26 COSY spectra
- 9 NOESY spectra
- 41 31P spectra
- 53 19F spectra
156 applications were carried out which jointly took 1074 hours of measurements.