D.S. Bolotin, M.Ya. Demakova, A.S. Novikov, M.S. Avdontceva, M.L. Kuznetsov, N.A. Bokach, V.Yu. Kukushkin
“Bifunctional Reactivity of Amidoximes Observed upon Nucleophilic Addition to Metal-Activated Nitriles”
Inorg. Chem., 2015, 54, 4039-4046
DOI:10.1021/acs.inorgchem.5b00253
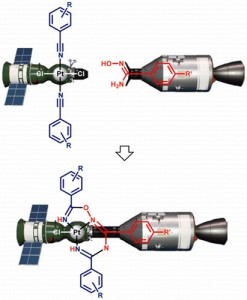
Treatment of the aromatic nitrile complexes trans-[PtCl2(RC6H4CN)2] with the aryl amidoximes p-R′C6H4C(NH2)═NOH, followed by addition of 1 equiv of AgOTf and then 5 equiv of Et3N, leads to the chelates [PtCl{HN═C(RC6H4)ON═C(C6H4R′-p)NC(RC6H4)═NH}] (15 examples; yields 71−88% after column chromatography) derived from the platinum(II)-mediated coupling between metal-activated nitriles and amidoximes. The combined experimental and theoretical results indicate that the coupling with the nitrile ligands involves both the HON and monodeprotonated NH2 groups of the amidoximes.