Ludmila L. Rodina, Jury J. Medvedev, Olesia S. Galkina and Valerij A. Nikolaev
“Thermolysis of 4-Diazotetrahydrofuran-3-ones: Total Change of Reaction Course Compared to Photolysis”
European Journal of Organic Chemistry 2014, 14, 2993–3000
DOI: 10.1002/ejoc.201400161
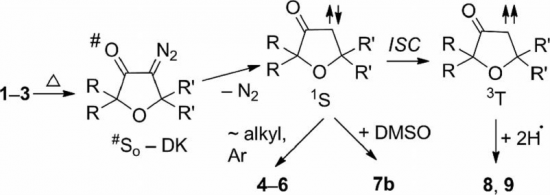
Thermolysis of 2,2,5,5-tetrasubstituted 4-diazodihydrofuran-3-ones in protic (BnOH) and aprotic (DMSO) media, in contrast to photolysis, gives rise to the formation of 2,2,4,5-substituted 3(2H)-furanones as a result of 1,2-alkyl (aryl) shift. The thermal stability of diazodihydrofuranones in these processes is determined by the nature of the substituents at the C-5 atom of the heterocyclic structure: alkyl and electron-donating groups in the para-position of the aryl substituent notably reduce the persistence of diazodihydrofuranones, whereas aryl groups and electron-withdrawing substituents increase their stability considerably. Thermolysis of substituted 4-diazodihydrofuran-3-ones in DMSO solution is a first-order reaction that furnishes higher yields of furanones than thermolysis in BnOH. The reaction can serve as a preparative method for the synthesis of tetrasubstituted-3(2H)-furanones.