A.A. Melekhova, A.S. Novikov, K.V. Luzyanin, N.A. Bokach, G.L. Starova, V.V. Gurzhiy, V.Yu. Kukushkin
“Tris-isocyanide copper(I) complexes: Synthetic, structural, and theoretical study”
Inorg. Chim. Acta, 2015, 434, 31-36
DOI:10.1016/j.ica.2015.05.002
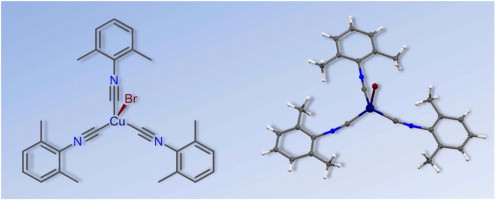
The reaction of CuBr with 3 equiv of CNR in CHCl3 at RT furnished the pure tris-isocyanide complexes [CuBr(CNR)3] [R = 2,6-Me2C6H3 (Xyl) (1), 2-Cl-6-MeC6H3 (2), 2-Naphthyl (3), C6H11 (Cy) (4)] in 85–99% isolated yield. Compounds 1–4 were characterized by elemental analyses (C, H, N), high resolution ESI+-MS, IR, 1H and 13C{1H} NMR spectroscopic techniques, and by single-crystal X-ray diffraction for 1 and 2. Coordination polyhedra of these two complexes are intermediate between tetrahedral and trigonal pyramidal with three isocyanide and one bromide ligands; the fragment M–C–N is bent [171.2(2)–175.6(2)°]. The influence of crystal packing effects on the geometric parameters of 1 as well as nature of the coordination Cu–C bonds and bond order of the formal triple C (triple bond)N bonds in this compound were also studied theoretically at the DFT level of theory. The crystal packing effects noticeably affect the values of the Cu–C–N angles and lead to a decrease of these angles approximately 10° relative to the gas phase geometries. Results of the AIM, NBO, and CDA analyses reveal that the Cu–C coordination bonds are of the electrostatic nature. Electrostatic interaction, unlike the covalent one, has no orientation, and it is the reason of the curved shape of the metal–ligand fragment.